How to Get Liters From Moles
For example in a 25 milliliter solution there are 000056 moles of OH-. N number of moles R gas constant 008206 L atmmol K T temperature in Kelvins P absolute pressure in atm V volume in liters P nRTV 8303e-18 008206 298 1 203e-16 atm.
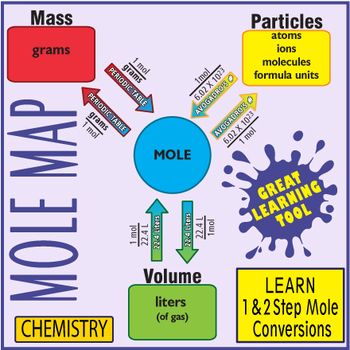
My Chemistry Students Find Using This Map The Easiest Way To Quickly Master 1 And 2 Step Mole Conversi Teaching Chemistry Chemistry Lessons Chemistry Education
In this case five liters times ten millimoles per liter.

. Add that to a container and bring the volume to five liters with water. If your pot contains 22 liters of water and we assume the density of the water is 1gmL then the mass of the water in the pot is 22 x 10 3 grams. To calculate the molarity of a solution the number of moles of solute must be divided by the total liters of solution produced.
The concentration of each product is found by dividing the number of moles of each ion over the total liters of solution created. Divide 000056 moles over 0025 liters to get the concentration OH- as 00225 moles per liter. First the molar mass allows you to change mass into mol.
If the amount of solute is given in grams we must first calculate the number of moles of solute using the solutes molar mass then calculate the molarity using the number of moles and total volume. Then multiply that by the molecular weight of 15756 grams per mole and you get seven point eight eight grams. We can now use the 407 kJmol factor to show that it will take 1222 mol x 407.
This will give you fifty millimoles or five times ten to the negative two moles. 2 If I have an unknown quantity of gas at a pressure of 12 atm a volume of 31 liters and a temperature of 87 0C how many moles of gas do I have. Comment by the ChemTeam.
Here I simply looked up the value of R rather. The molecular weight of water is 180 gmol so this means we have 22 x 10 3 grams180gmol 1222 moles of water. I will often change the pressure to atm so as to use the 008206 value.
Then knowing that there are 6022x1023 molecules in one mol Avogadros number you can find the number of molecules in however many mol of the substance that you have. Plug the values into the formula. Conversion from grams to molecules requires two conversion factors.
3 If I contain 3 moles of gas in a container with a volume of 60 liters and at a. 1 If I have 4 moles of a gas at a pressure of 56 atm and a volume of 12 liters what is the temperature.

Chemistry Triangle Conversion Tool Moles Liters Molarity Atoms Chemistry Medicine Notes High School Science

Molarity Dilution Problems Solution Stoichiometry Grams Moles Liters Volume Calculations Chemistry Youtub Math Facts Addition Chemistry Chemistry Classroom

Liters To Moles Conversion How To Convert Liters To Moles By Wayne Breslyn Mole Conversion Mole Converter

Molarity 2 Molarity M This Is The Most Common Expression Of Concentration M Molarity Moles Of Solute Mol Liter Study Chemistry Biology Notes Chemistry
No comments for "How to Get Liters From Moles"
Post a Comment